Bulletin #4338, Do Food Sterilizers Work?
Food Safety Facts
Bulletin #4338, Do Food Sterilizers Work? (PDF)
Developed by Kristi Crowe, Ph.D. Candidate, Department of Food Science & Human Nutrition, and Alfred Bushway, Extension Food Science Specialist
For information about UMaine Extension programs and resources, visit extension.umaine.edu.
Find more of our publications and books at extension.umaine.edu/publications/.
Home-use ozonation systems are being marketed to consumers as safe, effective methods of sterilizing produce. Should you buy one?
While proper cooking can prevent food-borne illness, fruits and vegetables are often eaten raw. Eating contaminated produce can lead to food-borne illness, which can be serious—and sometimes fatal.
Ozone, which in the upper atmosphere protects the Earth from ultraviolet radiation, is receiving increased attention as a sterilizer for fresh produce.
Ozone is a gas with three atoms of oxygen, compared to the two in the oxygen we breathe. Although ozone has been used in European water treatment facilities for nearly 100 years, the U.S. food industry has only recently expressed interest in this oxidizing agent. Ozone offers many advantages over traditional disinfectants. Compared to chlorine, the most widely used sanitizer for fresh produce, ozone has stronger oxidizing and antimicrobial activity. This makes it a good natural purifier. Further-more, ozone disinfects without producing toxic or cancer-causing compounds like those produced by chlorine treatments.
As an alternative to chlorine for removing microbes such as bacteria and mold from fresh fruits and vegetables, ozone may offer promising results for industrial food processing. But do you need an ozone food sterilizer at home?


Our evaluation
At the University of Maine, we evaluated two home-use food sterilzers — “ozone system A” and “ozone system B”1 — using lowbush blueberries. We measured the concentration of ozone produced by each unit, as well as each unit’s effectiveness in reducing the amount of bacteria and other microbes on the blueberries.
In our evaluation, blueberry samples were treated with ozonated water, distilled water, and chlorine.2 Two methods of ozone application were used:
- Blueberries were treated (according to product directions) by soaking them in two liters of distilled water with ozone gas bubbled through it, for a contact time of ten minutes. (Figure 1.)
- Blueberries were washed in two liters of ozonated distilled water for a contact time of two minutes. (Figure 2.)
We then analyzed microbe levels on the blueberries using FDA Standard Methods.3 We also measured ozone concentrations in the ozone washes using an indigo colorimetric method.4 Unwashed blueberries served as the control for this study.
| Figure 1. Microbes on blueberries treated with two home-use ozone-based food sterilizers. | Figure 2. Comparison of the microbes on blueberries treated with ozone, chlorine, and distilled water. |
|---|---|
Ten-minute ozone bubble-wash treatments. 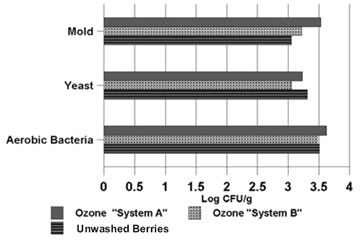 Figure 1 shows the microbes found on lowbush blueberries following treatment with home-use food sterilizers that bubbled ozone through two liters of distilled water for ten minutes. Bacteria and yeast populations on the berries treated with ozone system B were lower than the populations on the unwashed berries, but not significantly. Bacteria and mold counts on berries treated with ozone system A were actually higher than on the unwashed berries, and mold counts were higher in samples treated with system B. (The increase in microbial counts following treatment may be explained by variations arising from sampling.) | Two-minute wash treatments. 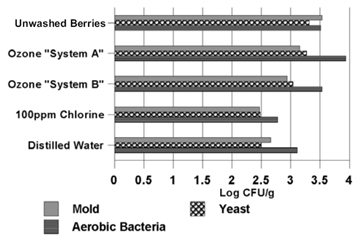 Figure 2 compares the antimicrobial effectiveness of two-minute washes using water ozonated with system A and system B6, distilled water, and chlorine (100 ppm). Berries treated with chlorine and distilled water had fewer microbes than berries treated with either of the ozone-based food sterilizers. Berries treated with chlorine had the fewest microbes. However, microbe populations were only marginally greater on berries treated with distilled water. Berries treated with water from ozone system A had more yeast and mold than berries treated with water from ozone system B. But both ozone systems produced berries with higher bacterial populations than the unwashed berries. (This may be explained by variations arising from sampling.) |
Our conclusion: distilled water is effective and affordable
Consumption of toxic residues from using the ozone washes did not prove to be a concern. After each trial we measured the concentration of ozone in the water. Residual ozone concentrations in the wash water following treatment were negligible — less than 0.001 mg of ozone per liter.
Home-use ozone system B killed more microbes than system A in both of our trials, although the differences were not statistically significant. The results of these trials reflect the variability and unpredictability of these two home-use ozonation systems.
Overall, home-use ozone systems did not improve food safety quality appreciably5 in our study, when they were used according to product directions. Considering these results, as well as the cost of such systems, we recommend washing your fresh fruits and vegetables with distilled water for 60 to 120 seconds as a more efficient and economical way to improve quality and safety.
1 “Ozone system A” = the Ozone Water Purifier XT-301 from Air-Zone, Inc., Leesburg, VA “Ozone system B” = the J0-4 Multi-Functional Food Sterilizer from Indoor Purification Systems, Layton, UT.
2 100-parts-per-million (ppm) chlorine solution
3 FDA. 1998. Bacteriological Analytical Manual, 8th ed. Association of Official Analytical Chemists. Maryland: AOAC International.
4 Bader H, Hoigne J. 1981. “Determination of ozone in water by the indigo method.” Water Res. 15: 449-456.
5 The initial microbial population was reduced by less than 0.5 log.
6 Ozone treatments for this trial were conducted by placing a 350-gram blueberry sample into two liters of ozonated distilled water for a contact time of 2 minutes.
Information in this publication is provided purely for educational purposes. No responsibility is assumed for any problems associated with the use of products or services mentioned. No endorsement of products or companies is intended, nor is criticism of unnamed products or companies implied.
© 2006
Call 800.287.0274 (in Maine), or 207.581.3188, for information on publications and program offerings from University of Maine Cooperative Extension, or visit extension.umaine.edu.
In complying with the letter and spirit of applicable laws and pursuing its own goals of diversity, the University of Maine System does not discriminate on the grounds of race, color, religion, sex, sexual orientation, transgender status, gender, gender identity or expression, ethnicity, national origin, citizenship status, familial status, ancestry, age, disability physical or mental, genetic information, or veterans or military status in employment, education, and all other programs and activities. The University provides reasonable accommodations to qualified individuals with disabilities upon request. The following person has been designated to handle inquiries regarding non-discrimination policies: Director of Institutional Equity and Title IX Services, 5713 Chadbourne Hall, Room 412, University of Maine, Orono, ME 04469-5713, 207.581.1226, TTY 711 (Maine Relay System).

